Molecular Aspects of Disease
Spring 2004
“Protein Misfolding and Ageing: Role in Neurodegeneration”
Based on a lecture given by Igor Bronstein, National
Institute of Animal Health
Contents:
Title page: 47 words
Text: 8 pages, 2000 words
References: 1 page, 705 words
Figures: 2, 127 words
Protein Misfolding in Neurodegeneration
A number of aetiologically distinct neurodegenerative disorders associated with ageing, e.g., Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), the tauopathies, and prion-related transmissible spongiform encephalopathies (TPE) such as Creutzfeldt-Jacob disease (CJD), show characteristic patterns of neuron damage due to deposition of protein aggregates in the brain1,2). These neurodegenerative diseases primarily involve the central nervous system (CNS) and show toxic abnormal protein deposition leading to neuronal cell death. The deposits are generally comprised of dense intra- or extracellularly located fibrils or plaques in which the protein molecules have a high percentage of β-pleated sheet secondary structure. The deposits are generally ubiquitinated but have survived proteosome targeting and often consist of a number of different cellular proteins. Neuronal cell death that occurs due to these deposits and the resultant neuroinflammatory response has a variety of pathological consequences in patients, including dementia (AD), parkinsonism, memory loss, and eventually death. As these neurodegenerative disorders show similar characteristics with regard to aberrant protein deposition in the CNS, further characterization of the normal cellular mechanisms for disposal of such deleterious misfolded proteins may facilitate the development of novel methods for their prevention and/or treatment.
Neurodegenerative Diseases
Alzheimer’s Disease
Alzheimer’s disease (AD), characterized by accumulation of extracellular plaques and cytoplasmic neurofibrillary tangles (NFTs)2), is the most common of these neurodegenerative diseases, affecting ~4 million people in the USA with annual AD-associated costs exceeding US$100 billion3), and almost 50% of the population over the age of 85 in both Europe and the US4,5). The major component of the dense plaques in AD is β-amyloid (βA), a peptide of ~40 amino acids derived from the larger amyloid precursor protein (APP). Accumulation of βA occurs early in the development of AD generally occurring many years prior to other brain lesions and symptoms. NFTs are bundles of helical filaments containing highly phosphorylated and ubiquitinated forms of the microtubule-associated factor tau6). AD primarily affects neurons in the hippocampus and basal forebrain, leading to brain shrinkage, and loss of both memory and intellect.
Tissue-specific processing has been shown to result in heterogeneity of the βA peptide at both its N and C termini7). Both β- and γ-secretase have been shown to cleave APP, but this cleavage is unlikely to be the primary pathological alteration in AD as the peptides thus produced are found in deposits and in other cells8). βA accumulation is considered to play a central role in the pathogenesis of AD, with preamyloid deposits increasing in density over time and eventually developing into neurotoxic amyloid plaques and neurofibrillary tangles9).
Parkinson’s Disease
Parkinson’s disease (PD), which has an incidence of 1.8% of the population over the age of 65 increasing from 0.6% from 65 to 69 years to 2.6% from 85 to 89 years10), mainly affects dopaminergic neurons in the substantia nigra-striatum pathway. Up to 70% of nigral cells are lost in PD, resulting in disruption of motor control pathways and progressive loss of control of body movement. PD patients show characteristic tremors, akinesia, bradykinesia, postural instability, and inability to control the extent of their movements11). These symptoms are thought to be due to the degeneration of dopaminergic neurons and the resulting abnormalities in dopamine levels. In addition to neuronal degeneration, the PD brain also contains ubiquitinated intracytoplasmic inclusion bodies known as Lewy bodies12). The major component of the Lewy bodies is α-synuclein, but immunohistochemical analyses have shown that they also contain ubiquitin and β-amyloid precursor protein13). Two different α-synuclein missense mutations have been found in dominant cases of PD both of which result in accelerated aggregate formation as compared with wild-type α-synuclein in vitro14).
The accelerated aggregation induced by these mutations suggests that PD patients have inefficient clearance of the aggregates by the proteasome.
Transmissible spongiform encephalopathies
The transmissible spongiform encephalopathies (TPEs) are a group of prion-related fatal neurodegenerative infectious disorders for which no effective treatment strategies have yet been developed. These disorders are characterized by the accumulation of a conformational variant of the normal cellular prion protein (PrPc)15,16). The infectious prion is an insoluble proteinaceous infectious particle with no nucleic acid component, which consists only of a modified isoform of PrP, designated PrPsc for “scrapie”, the ovine disease in which it was first identified. TPEs are unique in that the prion particle is itself transmissible17). Human TPEs include Creutzfeldt-Jakob disease, Gerstmann-Straussler-Scheinker disease, Kuru, and fatal familial insomnia, all of which are associated with pathological findings of spongiform vacuolation, gliosis, and neuronal cell loss. Although mutations have been found throughout the PrP protein, some mutations occur adjacent to elements of putative secondary structure15,18). These mutations may alter the structure of the protein, and thus affect its properties. Indeed, pathogenic PrPsc is a highly insoluble and protease-resistant structural isoform with reduced α-helix/increased β-sheet content, indicating that secondary/tertiary structural variations can result in marked functional differences in the protein.
Little is known about the aetiology of TPEs, the natural function of PrPc in healthy cells, or the mechanisms of transmissibility and pathogenesis of PrPsc. However, structural analyses have identified several domains of interest in the PrP molecule, which is encoded on the short arm of chromosome 20 in humans. As shown in Fig. 1, the N-terminus of the molecule contains five putative Cu binding domains, suggesting a role as a metal-containing sensor, scavenger, or DNA binding protein in normal neurons. In fact, Cu binding has been suggested to inhibit prion infectivity19). PrPc appears to have a role in neuroprotection associated with ageing and recovery from brain injury, as lack of PrPc has been shown to have pathological effects in aged animals and to impair recovery following traumatic brain injury20). These results suggest that loss of functional PrPc is involved in the pathogenesis of prion diseases. Its rapid recycling, specific proteolysis, and significant conformational change on cleavage support a role of PrPc as a multifunctional sensor molecule. The normal molecule is mostly α-helical in structure, while pathological PrPsc has a high percentage of β-sheet.
| Figure 1 |
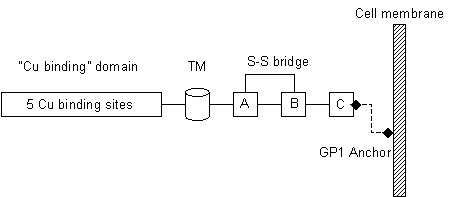 |
| Schematic representation of the PrP molecule. The N-terminus contains 5 putative Cu binding domains. The potential transmembrane domain (TM) is buried in the native conformation, and the molecule binds to the cell membrane via a GP1 anchor. The TM domain may be exposed in the pathological variant isoform PrPsc. The molecule contains several Cys residues, with a disulphide bridge between the A and B domains as shown in the native conformation. |
Ageing
From the perspective of evolutionary biology, it seems unlikely that there would be genes involved in ageing as few animals live long enough in the wild and generally only young adults are engaged in reproduction. Selection acts on “somatic maintenance”, i.e., tissue and cell regeneration and recovery from injury/insult, etc., and the organism can afford only limited investment in such maintenance resulting in gradual accumulation of cellular damage. However, most cells in the body are relatively short-lived, and do not persist long enough to accumulate major damage within the organism’s reproductive lifespan. Therefore, there would appear to be no mechanism for selective pressure to act on ageing. However, in contrast to the short-lived nature of somatic cells, neurons last throughout the animal’s lifetime and do accumulate injury with age, often leading to their degeneration and consequently to disease. Counterintuitively, the insight provided by post-genomic biology – in silico experiments, expression of human transgenes in animal models etc. – has revealed that there are genes involved specifically in ageing. Mutations in the genes associated with these diseases result in abnormal processing and accumulation of misfolded protein in neuronal inclusion bodies and plaque.
Experiments in the nematode Caenorhabditis elegans have identified genes that confer longevity. The dauer mutant larva is a non-feeding, developmentally arrested dispersal stage induced in nature under adverse environmental conditions. A number of mutations have been isolated in genes that affect the process of dauer formation, and these genes have been ordered in a genetic pathway. Dauer mutant larvae have a mutation in the daf2 gene, the product of which is similar to the human insulin GF1 receptor and shows homology with components of mammalian and Drosophila protein growth factor-mediated signal transduction pathways. These C. elegans mutants are long-lived, with a lifespan approximately double that of the wild-type adult21). This is of interest with regard to the role of protein misfolding in ageing and neurodegenerative diseases as the DAF2 receptor is part of an environmental sensing mechanism that begins a signal transduction cascade, which activates intracellular antioxidants and the heat shock proteins (HSPs). The HSPs are well-known to play a role in prevention of the protein misfolding associated with the ageing-related neurodegenerative diseases, as described above, as well as in diabetes. The similarity between DAF2 and the GF1 insulin receptor is also significant not only for this last point, but also with regard to the phenotypic similarity between dauer mutants and the results of calorie-restricted feeding, which has been shown to retard the rate of ageing in both mammals and invertebrates22,23).
Current knowledge about the biology of the diseases
Abnormal protein aggregation and deposition, caused by misfolding, have been shown to play important roles in the pathogenesis of a wide range of important ageing-related neurodegenerative disorders. The presence of protein aggregates in the brain suggests that these diseases share dysfunction of the molecular chaperone systems normally responsible for prevention of protein misfolding. Neurons are vulnerable to the toxic effects of mutant or misfolded protein as they are not subject to somatic maintenance and turnover. Dysfunction of the systems involved in detection and elimination of misfolded proteins, such as the HSPs and the ubiquitin-proteosome system (UPS) plays a role in the pathogenesis of neurodegenerative diseases. The UPS itself has also been suggested to be disrupted by toxic proteins in what essentially amounts to a catastrophic feedback mechanism leading to accumulation of ever-increasing amounts of abnormal protein24).
Misfolded protein deposition
A single polypeptide can adopt any one of a number of 3D conformations, usually only one of which is the correct ‘native’ state. Any other conformations that may form during the course of folding of the nascent polypeptide chain, such as folding intermediates, or as the result of insult (heat/redox/chemical/irradiation etc.) may be deleterious to the cell. The HSPs act as molecular chaperones and in regulating protein folding, and non-native misfolded proteins are targeted with ubiquitin for destruction by the proteosome. However, accumulation of excess amounts of misfolded protein due to overproduction, insult, or dysfunction of the chaperone system can lead first to prefibril formation then plaque and consequently to cell death and disease (Fig. 2).
| Figure 2 |
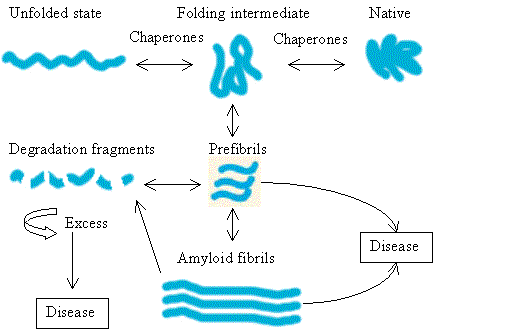 |
| Protein folding involves transition through folding intermediates stabilized by molecular chaperones. Protein misfolding can occur, resulting in prefibril formation. Normally, the prefibrils are degraded but under pathological conditions they may accumulate and lead to disease through production of amyloid fibrils. Production of excessive amounts of degradation fragments can also lead to disease. |
Future prospects
Further understanding of the mechanisms involved in disposal of aberrant proteins and of the effects of accumulation of such proteins on neuronal survival may allow rational drug design strategies for these diseases. Many neurodegenerative diseases are intimately associated with the mechanisms of ageing in general, such as the accumulation over time of active oxygen and nitrogen species. This oxidative stress leads to protein damage in the cell resulting in their accumulation and eventually to cell death through apoptosis. There is evidence that this process can be arrested somewhat by antioxidants, suggesting the potential benefit of antioxidant-based regimens.
It is important to determine the detailed mechanisms underlying the toxicity of mutant or misfolded proteins in these diseases. Future studies should seek to determine whether the neurotoxicity lies with the monomers, oligomers, or aggregates, the reasons for protein aggregation, the determinants of target cells, and the primary targets of the toxic proteins. Although there are differences between these diseases, their shared feature of aberrant protein deposition in the brain suggests that it may be possible to develop common therapeutic methodologies applicable across multiple disorders.
It may be possible to prevent expression of the aberrant protein or to accelerate its degradation.
Indeed, Morgan et al. reported that clearance of β-amyloid by vaccination with a βA peptide prevented memory loss in an animal model of Alzheimer’s disease25).
Other therapeutic strategies include inhibition of protein aggregation and blockade of neuronal apoptosis.
In the treatment of these diseases, it may be beneficial to up-regulate the expression of HSPs that protect against the toxic effects of misfolded protein. In addition, as the chaperones function in a cooperative manner in vivo, it may be necessary to increase the expression of multiple chaperones to obtain beneficial clinical effects. However, excessive chaperone levels have been reported to have deleterious effects and therefore great care will be necessary in evaluating the potential ill effects of any such methodologies designed to increase HSP levels prior to use in human trials.
References
- Paulson, H. L. Protein fate in neurodegenerative proteinopathies: polyglutamine diseases join the (mis)fold. (1999) Am. J. Hum. Genet. 64, 339-345
- Tolnay, M., and Probst, A. REVIEW: tau protein pathology in Alzheimer's disease and related disorders (1999) Neuropathol. Appl. Neurobiol. 25, 171-187
- Chapman PF, Falinska AM, Knevett SG, and Ramsay MF. (2001) Trends in Genetics. May;17(5):254-61.
- Launer LJ, Andersen K, Dewey ME, Letenneur L, Ott A, Amaducci LA, Brayne C, Copeland JR, Dartigues JF, Kragh-Sorensen P, Lobo A, Martinez-Lage JM, Stijnen T, Hofman A. Rates and risk factors for dementia and Alzheimer's disease: results from EURODEM pooled analyses. (1999) Neurology. Jan 1;52(1):78-84
- Fratiglioni L, Launer LJ, Andersen K, Breteler MM, Copeland JR, Dartigues JF, Lobo A, Martinez-Lage J, Soininen H, Hofman A. Incidence of dementia and major subtypes in Europe: A collaborative study of population-based cohorts. Neurologic Diseases in the Elderly Research Group. (2000) Neurology. 54(11 Suppl 5):S10-5.
- Morishima-Kawashima, M., Hasegawa, M., Takio, K., Suzuki, M., Titani, K., and Ihara, Y. Ubiquitin is conjugated with amino-terminally processed tau in paired helical filaments. (1993) Neuron. 10, 1151-1160
- Wisniewski, T., Ghiso, J., and Frangione, B. Biology of A beta amyloid in Alzheimer's disease. (1997) Neurobiol. Dis. 4, 313-328
- Selkoe, D. J. Cell biology of the amyloid beta-protein precursor and the mechanism of Alzheimer's disease. (1994) Annu. Rev. Cell Biol. 10, 373-403
- Hardy, J. New insights into the genetics of Alzheimer's disease. (1996) Ann. Med. 28, 255-258
- de Rijk M.C., Launer L.J., Berger K., Breteler M.M., Dartigues J.F., Baldereschi M., Fratiglioni L., Lobo A., Martinez-Lage J., Trenkwalder C., and Hofman A. Prevalence of Parkinson's disease in Europe: A collaborative study of population-based cohorts. Neurologic Diseases in the Elderly Research Group. (2000) Neurology. 54(11 Suppl 5):S21-3.
- Nussbaum, R. L., and Polymeropoulos, M. H. Genetics of Parkinson's disease (1997) Hum. Mol. Genet. 6, 1687-1691
- Duvoisin, R. C. Recent advances in the genetics of Parkinson's disease. (1996) Adv. Neurol. 69, 33-40
- Takeda, A., Mallory, M., Sundsmo, M., Honer, W., Hansen, L., and Masliah, E. Abnormal accumulation of NACP/alpha-synuclein in neurodegenerative disorders. (1998) Am. J. Pathol. 152, 367-372
- Narhi, L., Wood, S. J., Steavenson, S., Jiang, Y., Wu, G. M., Anafi, D., Kaufman, S. A., Martin, F., Sitney, K., Denis, P., Louis, J.-C., Wypych, J., Biere, A. L., and Citron, M. Both familial Parkinson's disease mutations accelerate alpha-synuclein aggregation. (1999) J. Biol. Chem. 274, 9843-9846
- Prusiner, S. B., Scott, M. R., DeArmond, S. J., and Cohen, F. E. Prion protein biology. (1998) Cell 93, 337-348
- DeArmond SJ, and Prusiner SB. Perspectives on prion biology, prion disease pathogenesis, and pharmacologic approaches to treatment. (2003) Clin. Lab. Med. 23(1): 1-41.
- Horwich, A. L., and Weissman, J. S. Deadly conformations--protein misfolding in prion disease. (1997) Cell 89, 499-510
- DeArmond, S. J., Sanchez, H., Yehiely, F., Qiu, Y., Ninchak-Casey, A., Daggett, V., Camerino, A. P., Cayetano, J., Rogers, M., Groth, D., Torchia, M., Tremblay, P., Scott, M. R., Cohen, F. E., and Prusiner, S. B. Selective neuronal targeting in prion disease. (1997) Neuron 19, 1337-1348
- Hijazi N., Shaked Y., Rosenmann H., Ben-Hur T., and Gabizon R. Copper binding to PrPC may inhibit prion disease propagation. (2003) Brain Res. Dec 12;993(1-2):192-200.
- Hoshino S, Inoue K, Yokoyama T, Kobayashi S, Asakura T, Teramoto A, and Itohara S. Prions prevent brain damage after experimental brain injury: a preliminary report. (2003) Acta Neurochir Suppl. 86:297-9.
- Kenyon C, Chang J, Gensch E, Rudner A, and Tabtiang R. A C. elegans mutant that lives twice as long as wild type. (1993) Nature. Dec 2;366(6454):461-4.
- Merry B.J. Molecular mechanisms linking calorie restriction and longevity. (2002) Int. J. Biochem. Cell Biol. Nov;34(11):1340-54.
- Anisimov V.N. Insulin/IGF-1 signaling pathway driving aging and cancer as a target for pharmacological intervention. (2003) Exp. Gerontol. Oct;38(10):1041-9.
- Bence N.F., Sampat R.M., and Kopito R.R. Impairment of the ubiquitin-proteasome system by protein aggregation. (2001) Science. May 25;292(5521):1552-5.
- Morgan D., Diamond D.M., Gottschall P.E., Ugen K.E., Dickey C., Hardy J., Duff K., Jantzen P., DiCarlo G., Wilcock D., Connor K., Hatcher J., Hope C., Gordon M., Arendash G.W. A beta peptide vaccination prevents memory loss in an animal model of Alzheimer's disease. (2000) Nature. Dec 21-28;408(6815):982-5.